53rd Annual Meeting of the American Society of Clinical Oncology (ASCO)
 CHICAGO – In an early clinical trial, 33 out of 35 (94%) patients had clinical remission of myeloma upon receiving a new type of immunotherapy ̶ chimeric antigen receptor (CAR) T cells targeting B-cell maturation protein or BCMA. Most patients had only mild side effects. The study was presented at the 2017 American Society of Clinical Oncology (ASCO) Annual Meeting.
CHICAGO – In an early clinical trial, 33 out of 35 (94%) patients had clinical remission of myeloma upon receiving a new type of immunotherapy ̶ chimeric antigen receptor (CAR) T cells targeting B-cell maturation protein or BCMA. Most patients had only mild side effects. The study was presented at the 2017 American Society of Clinical Oncology (ASCO) Annual Meeting.
“Although recent advances in chemotherapy have prolonged life expectancy in multiple myeloma, this cancer remains incurable,” said study author Wanhong Zhao, MD, PhD, an associate director of hematology at The Second Affiliated Hospital of Xi’an Jiaotong University in Xi’an, China. “It appears that with this novel immunotherapy there may be a chance for cure in multiple myeloma, but we will need to follow patients much longer to confirm that.” Continue reading
53rd Annual Meeting of the American Society of Clinical Oncology (ASCO)
Chicago – According to Phase 3 ‘482 study, the largest international multiple myeloma trial ever conducted with 1,718 patients involved, patients who receive denosumab (XGEVA) had a significantly lower rate of renal adverse events compared to zoledronic acid (10.0 percent versus 17.1 percent, p<0.001). The study met its primary endpoint, demonstrating denosumab is non-inferior to zoledronic acid in delaying the time to first on-study skeletal-related event in patients with myeloma (HR=0.98, 95 percent CI: 0.85, 1.14; p=0.01). ). These results was presented yesterday at the 53rd Annual Meeting of the American Society of Clinical Oncology (ASCO). Continue reading
53rd Annual Meeting of the American Society of Clinical Oncology (ASCO)
June, 2, 2017
 CHICAGO – About 50% of all cancer survivors report moderate to high fear of recurrence. The fear can be so distressing that it negatively affects medical follow-up behavior, mood, relationships, work, goal setting, and quality of life, according to a study presented during the 53rd American Society of Clinical Oncology (ASCO) Annual Meeting taking place June 2-6 in Chicago. Yet, interventions to alleviate this fear are lacking. Continue reading
CHICAGO – About 50% of all cancer survivors report moderate to high fear of recurrence. The fear can be so distressing that it negatively affects medical follow-up behavior, mood, relationships, work, goal setting, and quality of life, according to a study presented during the 53rd American Society of Clinical Oncology (ASCO) Annual Meeting taking place June 2-6 in Chicago. Yet, interventions to alleviate this fear are lacking. Continue reading
 We all were saddened to hear of our recent loss of Yacob Yacobovitch, who passed away on the 18th of april 2017. Yacob Yacobovitch was member of Myeloma Patients Europe from 2012 until 2016. Continue reading
We all were saddened to hear of our recent loss of Yacob Yacobovitch, who passed away on the 18th of april 2017. Yacob Yacobovitch was member of Myeloma Patients Europe from 2012 until 2016. Continue reading
European Commission (EC) has granted approval daratumumab for use in combination with lenalidomide and dexamethasone, or bortezomib and dexamethasone, for the treatment of adult patients with myeloma who have received at least one prior therapy. Continue reading
The MPE Advocate Development Programme (ADP) is a training programme directed to MPE members with the aim to provide them with the skills and knowledge that any myeloma patient advocate should have regarding the drug development process. The main goal of MPE is to put medicines research into a myeloma context and create a tailored training programme for the myeloma community. Continue reading
 Patients suffering from rare blood disorders will benefit from networks of excellence across the European Member States.
Patients suffering from rare blood disorders will benefit from networks of excellence across the European Member States.
At the third conference on European Reference Networks (ERNs) this month in Vilnius, Lithuania, EuroBloodNet was officially announced as one of the recently approved ERNs. Continue reading
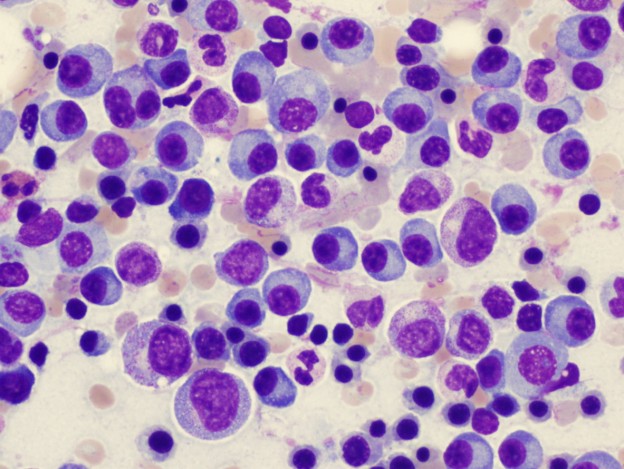 PHOENIX – Mayo Clinic researchers have found that an experimental drug, LCL161, stimulates the immune system, leading to tumor shrinkage in patients affected by myeloma. The findings are published in Nature Medicine.
PHOENIX – Mayo Clinic researchers have found that an experimental drug, LCL161, stimulates the immune system, leading to tumor shrinkage in patients affected by myeloma. The findings are published in Nature Medicine.
Myeloma is a blood cancer that affects plasma cells – white blood cells that normally produce antibodies to fight infection. Rather than produce helpful antibodies, the cancer cells, as they grow, secrete large amounts of a single antibody that accumulate in the body, causing kidney problems and infections. Continue reading
 It is with great sadness that we share with you that our friend, colleague and Board member Mika Peltovaara (23 Dec 1965 – 21 Febr 2017) passed away in the morning of the 21th of February 2017.
It is with great sadness that we share with you that our friend, colleague and Board member Mika Peltovaara (23 Dec 1965 – 21 Febr 2017) passed away in the morning of the 21th of February 2017.
Mika was board member of Myeloma Patients Europe since 2014. He was a very experienced, long-serving patient advocate for the Finnish Cancer Patient Association, with a special interest in possibilities of social media and new technologies in connecting with patients. He was a myeloma patient himself for many years. Continue reading
The contracts provided to patient advocates are often too long, difficult to understand for patient advocates, contain ambiguous clauses, contain terms that are in conflict with the very nature of patient advocacy, or even put the signing patient advocate at legal risk. That is what a survey run by the pan-European patient advocacy network Myeloma Patients Europe (MPE), and further supported by a network of 21 pan-European cancer patient advocacy organisations (WECAN) from other disease areas, shows. Continue reading
